Preparation Methods of Metal Powder | Industry Information

Preparation Methods of Metal Powder
Powder metallurgy is a process for preparing metal powders and using metal (or metal and non-metal mixture) powder as raw materials to form parts and products by molding and sintering. As the main raw material of the industry, metal powder is widely used in the fields of machinery, metallurgy, chemical industry, and aerospace materials. Metal powder is the basic raw material of the powder metallurgy industry, and its output and quality determine the development of the powder metallurgy industry. In this article, we will take a close look at the preparation methods of metal powder.

Preparation Methods of Metal Powder
The metal powder is usually an aggregate of metal particles of less than 1 mm, and there is no uniform regulation of the division of the particle size interval.
Commonly used methods are as follows: particles between 1000~50μm are conventional powder; 50~10μm is a fine powder; 10~0.5μm is a very fine powder; <0.5μm is an ultrafine powder; 0.1~100nm is nanopowder.
Each of the powder particles may be a crystal or a plurality of crystals depending on the particle size and preparation method.
Preparation Methods of Metal Powder
At present, there are dozens of methods for industrially producing powders. However, in terms of the actual analysis of the production process, it is mainly divided into mechanical and physical-chemical methods, which can be obtained from the direct refinement of solid, liquid, and gaseous metals, and the metal compounds in different states are obtained by reduction, pyrolysis, and electrolysis.
Carbides, nitrides, borides, and silicides of refractory metals can generally be obtained directly by compounding or reduction-combination methods. The shape, structure, and particle size of the same powder often vary greatly depending on the method of preparation.

Preparation Methods of Metal Powder
Preparation Methods of Metal Powder - 1. Mechanical Physics
The mechanical method is a processing way for breaking a metal into a powder of the desired particle size by means of a mechanical external force, and the chemical composition of the material in the preparation process is substantially unchanged.
The currently widely used methods are ball milling and grinding, which have the advantages of simple process and large yield, and can prepare ultrafine powders of high melting point metals and alloys which are difficult to obtain by conventional methods.
* Ball Milling
The ball milling method is mainly divided into a rolling ball method and a vibration ball milling method, which utilizes a mechanism in which metal particles are strained at different strain rates to break and refine.
This method is mainly suitable for the preparation of powders such as Sb, Cr, Mn, and Fe-Cr alloys. It has the advantages of continuous operation, high production efficiency, is suitable for dry grinding and wet grinding, and can be used for powder preparation of various metals and alloys.
The disadvantage is that the selectivity of the material is not strong, and it is difficult to classify during the powder preparation process.

TEM images of the powder samples were obtained by ball milling for 12h (a), 18h (b), and 24h (c) at 150r/min
* Grinding
The grinding method is to spray the compressed gas through the special nozzle and spray it into the grinding zone, thereby causing the materials in the grinding zone to collide with each other and rub into powder. After the airflow expands, the material rises into the classification zone, and the material of the particle size is sorted by the turbine classifier, and the remaining coarse powder is returned to the grinding zone to continue grinding until the required particle size is separated.
The grinding method is widely used in ultra-fine pulverization of non-metal, chemical raw materials, pigments, abrasives, health care drugs, and other industries. Since the grinding is produced by the dry method, the process of dehydration and drying of the material is omitted. The product has high purity, high activity, good dispersibility, fine particle size and narrow distribution, and smooth surface of the particles.
However, the grinding method has a high manufacturing cost, and in the production process of the metal powder, continuous inert gas or nitrogen gas must be used as a compressed gas source, and the gas consumption is large, so it is only suitable for crushing and milling of brittle metals and alloys.
* Atomization
The atomization is generally carried out by using a high-pressure gas or a high-pressure liquid or a blade rotating at a high speed to break a high-temperature, high-pressure molten metal or alloy into fine droplets, and then condensing in a collector to obtain an ultrafine metal powder.
The atomization method is one of the main methods for producing metal and alloy powder, and there is no chemical change in this process. The atomization method is generally applied to the production of metal powders such as Fe, Sn, Zn, Pb, Cu, etc., and can also be used for the production of alloy powders such as bronze, brass, carbon steel, and alloy steel.
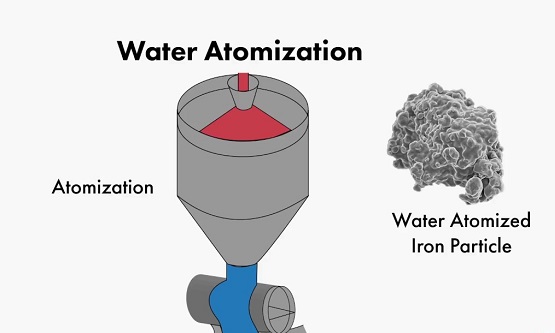
Atomization
The atomized powder has the advantages of high sphericity, controllable powder particle size, low oxygen content, low production cost, and adaptability to the production of various metal powders. It has become the main development direction of high-performance and special alloy powder preparation technology.
However, the atomization method has the defects of low production efficiency, low yield of ultrafine powder, and relatively large energy consumption.

The Microstructure of 3D-printed Stainless Steel Powder
Preparation Methods of Metal Powder - 2. Physical-chemical Method
The physicochemical method refers to a method for producing an ultrafine powder by changing the chemical composition or the state of aggregation of the raw material during the preparation of the powder. According to the different chemical principles, it can be divided into the reduction method, electrolysis method, and chemical replacement method.
* Reduction
The reduction is a method of reducing a metal oxide or a metal salt by using a reducing agent under certain conditions to obtain a metal or alloy powder and is one of the most widely used milling methods in production.
Commonly used reducing agents are gas-reducing agents (such as hydrogen, decomposing ammonia, converting natural gas, etc.), solid carbon-reducing agents (such as charcoal, coke, anthracite, etc.) and metal-reducing agents (such as calcium, magnesium, sodium, etc.).
Hydrogenation dehydrogenation with hydrogen as the reaction medium is the most representative preparation method. It utilizes the characteristics of easy hydrogenation of the raw material metal, hydrogenates the metal with hydrogen at a certain temperature to form a metal hydride, and then mechanically breaks the obtained metal hydride into a powder of the desired particle size, and then breaks the metal hydride. The hydrogen in the powder was removed under a vacuum to obtain the metal powder.
The reduction method is mainly applied to the preparation of metal (alloy) powders such as titanium, iron, tungsten, molybdenum, niobium, and tungsten-rhenium.
For example, titanium (powder) starts to react violently with hydrogen at a certain temperature. When the hydrogen content is more than 2.3%, the hydride is loose and easily pulverized into fine particles of hydride titanium powder. The titanium hydride powder is decomposed at a temperature of about 700 ° C and most of the hydrogen dissolved therein is removed to obtain the titanium powder.
The advantages of the reduction method are simple operation, easy control of process parameters, high production efficiency, low cost, and suitability for industrial production. The disadvantage is that it is only suitable for metal materials which are easy to react with hydrogen and become brittle and easily broken after hydrogen absorption.
* Electrolysis
Electrolysis is a method in which metal powder is deposited on a cathode by electrolytically melting an aqueous solution of a salt or a salt. The electrolytic aqueous solution can produce metal (alloy) powders such as Cu, Ni, Fe, Ag, Sn, and Fe-Ni, and the electrolytic molten salt can produce metal powders such as Zr, Ta, Ti, and Nb.
The advantage of the electrolysis method is that the purity of the prepared metal powder is relatively high, and the purity of the elemental powder can be up to 99.7% or more.
In addition, the electrolysis method can control the particle size of the powder well, and ultrafine powder can be obtained. However, electrolytic powdering consumes a large amount of electricity and has a high cost of milling.
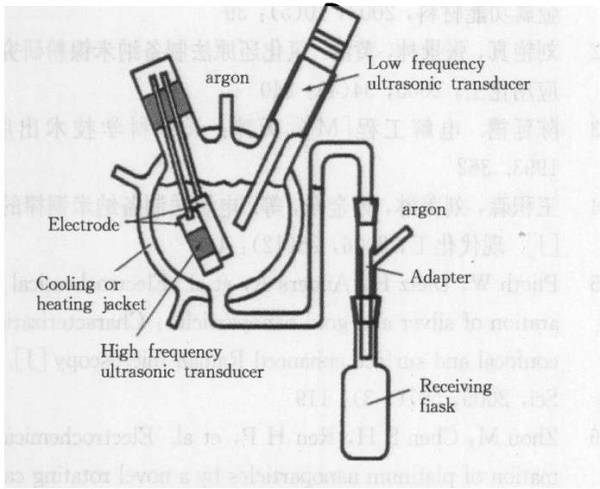
Ultrasonic Electrolysis Preparation of Iron Powder Device
* Hydroxyl
The hydroxyl method refers to the synthesis of certain metals (iron, nickel, etc.) and carbon monoxide into metal carbonyl compounds, which are then thermally decomposed into metal powders and carbon monoxide. Industrially, the hydroxyl method is mainly used to produce fine powders and ultrafine powders of nickel and iron, and alloy powders such as Fe-Ni, Fe-Co, and Ni-Co. The powder obtained by the hydroxyl method is very fine and has high purity, but the cost is also high.
* Chemical Replacement
The chemical replacement method is based on the activeness of the metal, and the metal has a small activity that is displaced from the metal salt solution by a highly active metal, and the metal obtained by the substitution is further processed and refined by other methods. This method is mainly applied to the preparation of inactive metal powders such as Cu, Ag, and Au.
Summary
Thank you for reading our article and we hope it can help you to have a better understanding of the preparation methods of metal powder. If you want to know more about metal powder, you can visit Advanced Refractory Metals (ARM) for more information.
Headquartered in Lake Forest, California, USA, ARM is one of the leading manufacturers & suppliers of metal powders around the world, providing people with high-quality refractory metal powders such as tungsten powder at a very competitive price.
{{item.content}}
LEVE A REPLY
{{item.children[0].content}}
{{item.content}}






