Niobium Used As A Photoactive Semiconductor

Introduction
As energy demand increases and environmental concern rises, people start to search for new renewable energy sources such as wind, solar, and geothermal power. Lots of advanced techniques including artificial photosynthesis and water-splitting reaction are developed to generate solar fuels. Moreover, photoactive semiconductor materials like Niobium are also utilized to enhance the efficiency of these processes.

Figure 1. Niobium
This article is going to explore these reactions and the niobium-based catalyst. Hope that you can have a better understanding of how we acquire solar fuel energy with niobium-based photoactive materials.
How Does Artificial Photosynthesis Happen?
In order to obtain solar energy, scientists got inspiration from Nature. Natural photosynthesis harnesses the power of sunlight and stores it in the form of glucose. This process takes place inside green plants and serves as the foundation of all living creatures. Natural photosynthesis also accounts for the formation of fossil coals. Artificial photosynthesis mimics the natural process and converts sunlight into hydrogen and other solar fuels. Such a process happens in photosynthetic cells and requires the assistance of photoactive catalysts.
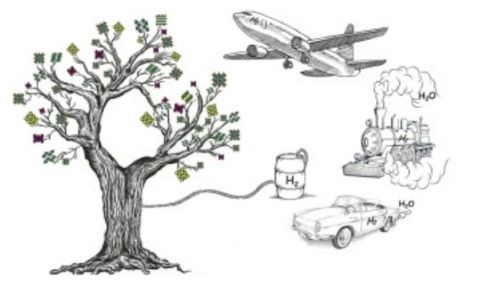
[1]
Figure 2. Artificial Photosynthesis
How Does Water Splitting Occur?
The synthetic reaction can be divided into hydrogen evolution and the photo-reduction of carbon dioxide. The first evolution part is also called water splitting in artificial photosynthesis. As for natural photosynthesis, water is oxidized, and oxygen and protons release consequently. Meanwhile, carbon dioxide is transformed into glucose. In terms of artificial photosynthesis, various water-splitting catalysts are employed, and the protons gained can be converted into hydrogen. In this way, clean, stable, and transportable hydrogen energy is produced efficiently.
The synthetic process has three steps.
- The first step is the harvest of light photons. Initially, only light at wavelengths of about 400-700 nm could be absorbed. Nowadays, a wider band in the solar spectrum can be utilized with the help of photo-catalysts.
- Next, transfer the charge generated to the surface of the catalysts.
- The last and most important procedure is the water-splitting reaction. Photo-electrochemical cells, semiconductor nanowires, and proton-conducting membranes are also needed.
Niobium-based Photoactive Water Splitting Catalyst
Niobium-based photoactive semiconductors have been extensively used as water-splitting catalysts because of the following features.
- Desirable bandgap: The photo-catalyst must have the desirable bandgap that allows oxidation and reduction reactions to happen. Namely, the catalyst should have a minimum bandgap of 1.23eV so that H2O would change into O2 and H+ would transform into H2.
- Positive conduction band: The valence band and conduction band matter. The more positive the conduction band is, the higher the reduction potential the catalyst has. For instance, niobates possess a more positive position of conduction bands compared with tantalates, so they have higher reduction potentials for redox reactions.
- Special structures: Niobium-based semiconductors have special structures, so they have better photocatalytic performance. Niobium compounds have different structures due to their crystallization. For instance, the symmetry of NbO4-tetrahedral and NbO6-octahedral units could be easily tailored, and Nb5+ has a large affinity to oxygen.
- Various types: Nb-based materials can be categorized into three types. Niobium pentoxide stands out for its electronic and morphological properties. Niobium-layered compounds are promising photo-catalysts with a 2D layered arrangement and a large surface area. You can add niobium as a dopant agent to improve the reaction efficiency as well.
Other Uses of Niobium
Niobium is a grey ductile metal with a range of applications.
About 90% of niobium is used to manufacture high-end steel. Adding a small amount of niobium to the steel could increase the strength, toughness, and corrosion resistance of steel. The weldability and formability could also be improved.
Niobium is commonly applied to make superconducting materials as well. The well-known Apollo Service Module, the Large Hadron Collider, and the International Thermonuclear Experimental Reactor employed numerous superconductors.
Related reading: 10 Important Uses Of Niobium
Conclusion
A variety of niobium-based photoactive semiconductors are used as catalysts to enhance the performance of artificial photosynthesis and solar water splitting because of their chemical and structural properties. Advanced Refractory Metals (ARM) is a leading supplier of niobium and its alloys. For more information, please check our homepage.
Reference:
[1] Celal Avcıoğlu, Suna Avcıoğlu, Maged F. Bekheet, Aleksander Gurlo (2022). Solar hydrogen generation using niobium-based photocatalysts: Design strategies, progress, and challenges. Materials Today Energy, 24. https://doi.org/10.1016/j.mtener.2021.100936.
{{item.content}}
LEVE A REPLY
{{item.children[0].content}}
{{item.content}}






